CMS Structure
CMS Oxygen has a larger atomic number than nitrogen, so one would expect this to be the bigger molecule. However, due to various electrical forces acting on the electron shell, the effective size of the nitrogen molecule is bigger than oxygen molecule. Under pressure, O2 selectively enters the pores at a higher rate than the N2. O2 leaves the pores during depressurization.
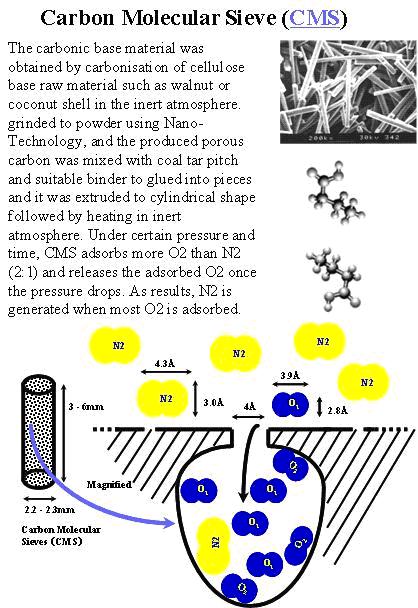
One angstom = 0.1 x 10-6 millimeters or 0.0001 um

Under pressure, more of the smaller, blue O2 molecules adhere to the pores than the larger, green N2 molecules. Enriched N2 continues out of the pressurized bed. After decompression, the O2 molecules vent to the atmosphere.
